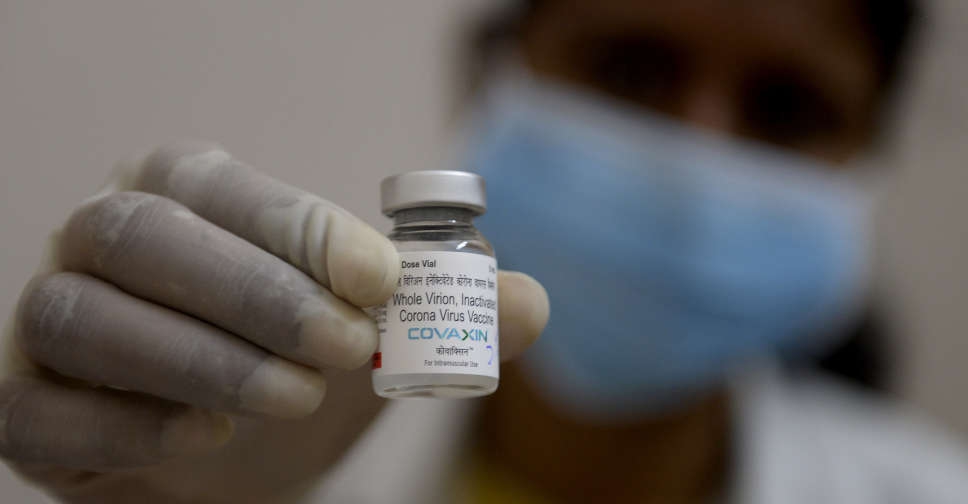
A World Health Organisation (WHO) technical advisory group was reviewing data on India's Covaxin shot against COVID-19 on Tuesday with a decision on its emergency use listing likely soon, a spokesperson said.
"If all is in place and all goes well and if the committee is satisfied, we would expect a recommendation within the next 24 hours or so," Margaret Harris told journalists at a UN press briefing.
Millions of Indians have taken the shot produced by Bharat Biotech but many have been unable to travel pending the WHO approval.



 Partial resumption of air navigation in Qatar
Partial resumption of air navigation in Qatar
 Kuwait and Qatar thwart fresh wave of Iranian attacks
Kuwait and Qatar thwart fresh wave of Iranian attacks
 After a week of war, Trump demands Iran's 'unconditional surrender'
After a week of war, Trump demands Iran's 'unconditional surrender'
 Ex-rapper's party set to sweep Nepal election
Ex-rapper's party set to sweep Nepal election
 Israeli airforce pounds Beirut, Lebanon death toll rises
Israeli airforce pounds Beirut, Lebanon death toll rises